Dehumidification for the aerospace industry
For the aerospace industry, reliable functional quality of all components is essential. Insufficient and excessive humidity can cause damage and defects to electronic components with potentially disastrous consequences. It affects the quality of polymerization of composites, sealants, coatings and welds, so that parts can deform, become porous or the quality of the surface is impaired.
Corrosion of steel on a drop of water

Iron cannot rust in the mere dry air. However, when moisture settles on the metal, oxygen (O2) and water (H2O) begin to react to form hydroxide ions (OH-). To balance the electrons required for this reaction, the iron oxidizes, i.e. it releases its electrons, which are then absorbed by the oxygen. This process produces iron oxide. Where the electrons have been withdrawn, an electron shortage occurs and positively charged iron atoms (Fe2+) are released. These migrate into the water droplets and combine there with the negatively charged hydroxide ions (OH-). In the first step, iron(II) hydroxide is formed as a result of different charges. Further reactions with water, oxygen and hydroxide ions result in ever more continuous reactions from which iron(III) oxide and iron(III) hydroxide are formed. They settle on the metal surface and give the rust its typical appearance. Unlike with metals such as aluminum, the process is only stopped when iron is no longer present.
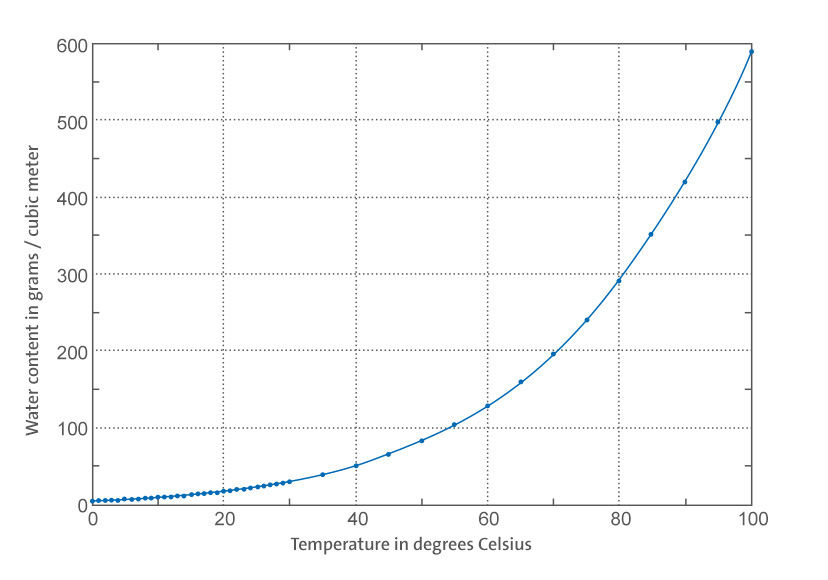
Saturation of water vapor in the air
Condair quality
Our customized systems offer professional solutions for air humidification and dehumidification for the size and conditions of each individual room. Because professional planning is the basis for functional quality and energy efficiency, we are happy to assist you right from the planning phase.

Szklarnie
Aby uzyskać optymalne plony i zminimalizować ryzyko inwazji szkodników, wilgotność w szklarniach musi być regulowana. Każdy gatunek rośliny ma swoją ...

Hammam, Caldarium i Rhassoul
Hamam to łaźnia parowa, w której odbywa się specjalna ceremonia kąpieli,
i jest ważną częścią islamskiej kultury kąpielowej i fizycznej.
Skupia się...

Łaźnia parowa i prysznic parowy
W dzisiejszych czasach wiele hoteli i klubów zdrowia oferuje zaplecze spa dla dobrego samopoczucia swoich gości i członków:
Basen, sauna, a coraz czę...

Steam bath and steam shower
Nowadays, many hotels and health clubs offer spa facilities for the well-being of their guests and members:
A swimming pool, a sauna and more and mor...

Centrum telefoniczne
Dobra wilgotność powietrza w call center zapewnia nie tylko dobre samopoczucie pracowników, lecz również ich wydajność i zdrowie.Choroby układu oddec...

Muzea
Muzea są skarbcem chroniącym dzieła sztuki i inny dorobek ludzkości. Niezliczone,
często unikalne i niezastąpione eksponaty, są warte wiele miliardó...

Magazynowanie
Wszędzie tam, gdzie przechowywane są produkty i towary lub archiwizowane są dokumenty, kompleksowe osuszanie powietrza chroni przed potencjalnym szko...

Pływalnie
Wysoki poziom parowania wody na pływalniach, szczególnie w połączeniu z panującymi tam wysokimi temperaturami w pomieszczeniu, prowadzi do ogromnego ...

Drukarnie
Nawet jeśli czytanie zaczyna wychodzić z mody, papier pozostaje cennym surowcem. Papier jest potrzebny nie tylko do gazet i książek, lecz także do pl...

Przemysł drzewny
Drewno to naturalny materiał, który „żyje” długo po ścięciu drzewa. Jeżeli podczas składowania i przetwarzania powietrze jest zbyt suche, drewno trac...